 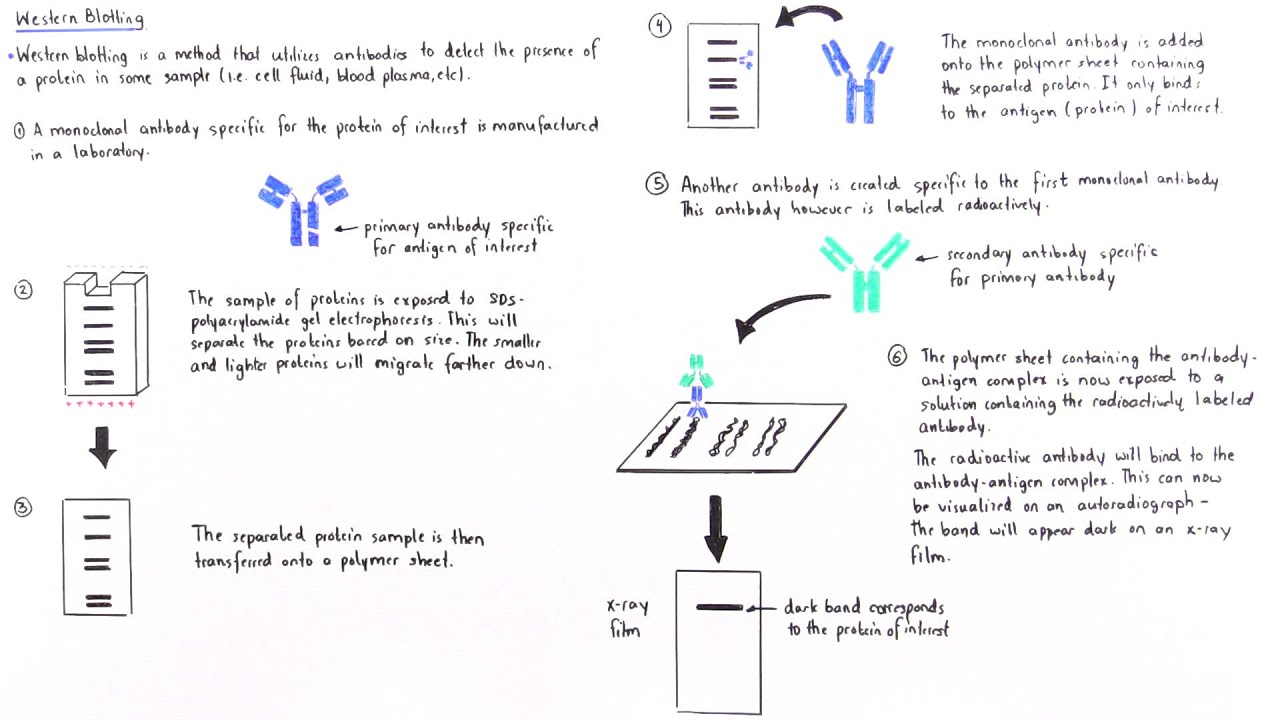
After binding of the enzyme–antibody conjugate, the addition of a chromogenic substrate that produces a highly colored and insoluble product causes the appearance of a colored band at the site of the target antigen. However, the most generally used detection procedures employ enzyme-linked antibodies against the protein. If the protein of interest is bound by a radioactive antibody, its position on the blot can be determined by exposing the membrane to a sheet of X-ray film, a procedure called autoradiography. The antigen-antibody complexes that form on the band containing the protein recognized by the antibody can be visualized in a variety of ways. The protein bands thus obtained are transferred onto a nitrocellulose or nylon membrane where they are “probed” with antibodies specific to the protein to be detected. It utilizes SDS-PAGE (Sodium dodecyl sulfate polyacrylamide gel electrophoresis), a type of gel electrophoresis to first separate various proteins in a mixture on the basis of their shape and size. Western blotting is usually done on a tissue homogenate or extract. The thickness of the band corresponds to the amount of protein present thus doing a standard can indicate the amount of protein present. As the antibodies only bind to the protein of interest, only one band should be visible. The bound antibodies are then detected by developing the film. The unbound antibody is washed off leaving only the bound antibody to the protein of interest. The membrane is then incubated with labels antibodies specific to the protein of interest. These results are then transferred to a membrane producing a band for each protein. In this technique, a mixture of proteins is separated based on molecular weight, and thus by type, through gel electrophoresis. In addition of substrate, a substrate reacts with the enzyme that is bound to the secondary antibody to generate colored substance, namely, visible protein bands. For detection of the proteins, primary antibody and enzyme-conjugated secondary antibody are used. Without pre-activation, proteins combine with nitrocellulose membrane based on hydrophobic interaction ( Blotting). Proteins are moved from within the gel onto a membrane made of Nitrocellulose (NC) or Polyvinylidene difluoride (PVDF). Marking target protein using a proper primary and secondary antibody to visualize (Detection).Įlectrophoresis used to separate proteins according to their electrophoretic mobility which depends on the charge, size of protein molecule, and structure of the proteins.Separation of proteins by size (Electrophoresis).The technique consists of three major processes: Western Blotting Technique Western blot (WB)or immunoblotis a workhorse immunoassay for most labs used to demonstrate antibody specificity, confirm gene expression, detect post-translational modifications (PTMs), diagnose diseases, and more. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
